Dean of Student Affairs
 In October 2022, Paul Kögerler was elected to serve as Dean of Student Affairs of RWTH's Faculty of Mathematics, Computer Science and Natural Sciences.
In October 2022, Paul Kögerler was elected to serve as Dean of Student Affairs of RWTH's Faculty of Mathematics, Computer Science and Natural Sciences.
How can we strengthen the notoriously weak magnetic coupling between bridged lanthanide ions?
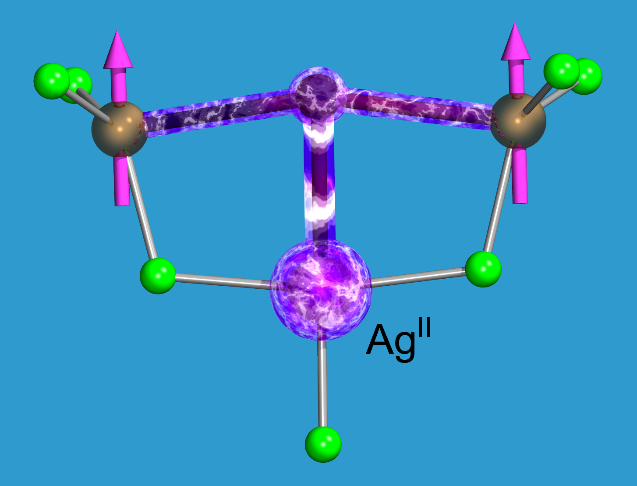 With their high magnetic moments (up to ca. 11 μB for Dy3+ or Ho3+) and their strong spin-orbit coupling, certain lanthanide ions should be highly attractive components of molecular magnetic materials and also molecular spintronics. At the same time, their spin-bearing 4f orbitals are contracted (read: teeny-weeny), which limits their overlap with the frontier orbitals of bridging ligands and, thus, the intramolecular magnetic exchange interactions. In turn, their highly interesting magnetic phenomena manifest only at very low temperatures. In the binational project POL-LAN-DE, funded by NCN (PL) and DFG (DE), our friends from Warsaw and us explored how the magnetic polarisation introduced by electron-hungry Ag2+ cations can salvage this situation. The Ag2+ cation has a 4d9 configuration and a 4d electron hole with an extremely high electron affinity of close to 11 eV, leading a pronounced hybridization with the valence orbitals of surrounding ligands. This induces a strong spin polarization even on rock-hard fluoride bridging ligands and can greatly increase the magnetic coupling in Ln-F-Ln bridges. For a systematic study, see
With their high magnetic moments (up to ca. 11 μB for Dy3+ or Ho3+) and their strong spin-orbit coupling, certain lanthanide ions should be highly attractive components of molecular magnetic materials and also molecular spintronics. At the same time, their spin-bearing 4f orbitals are contracted (read: teeny-weeny), which limits their overlap with the frontier orbitals of bridging ligands and, thus, the intramolecular magnetic exchange interactions. In turn, their highly interesting magnetic phenomena manifest only at very low temperatures. In the binational project POL-LAN-DE, funded by NCN (PL) and DFG (DE), our friends from Warsaw and us explored how the magnetic polarisation introduced by electron-hungry Ag2+ cations can salvage this situation. The Ag2+ cation has a 4d9 configuration and a 4d electron hole with an extremely high electron affinity of close to 11 eV, leading a pronounced hybridization with the valence orbitals of surrounding ligands. This induces a strong spin polarization even on rock-hard fluoride bridging ligands and can greatly increase the magnetic coupling in Ln-F-Ln bridges. For a systematic study, see
M. Domański, J. van Leusen, M. Metzelaars, P. Kögerler, W. Grochala, "Ag(II) as Spin Super-Polarizer in Molecular Spin Clusters", J. Phys. Chem. A 2022, 126, 9618-9626. ( doi:10.1021/acs.jpca.2c06032). doi:10.1021/acs.jpca.2c06032).
Cyclophanes based on pyrene and ferrocene for molecular spintronics
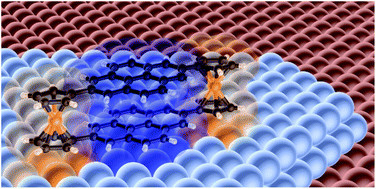 Cyclophanes providing two (or more) aromatic π systems are of great interest in molecular spintronics as one aromatic group can strongly chemisorb onto e.g. ferromagnetic metal substrate, leading to magnetic interfacial effects such as magnetic hardening, and imparting spin polarization on the remaining aromatic building blocks. We found that ferrocene pillars are able to lock two pyrene groups, i.e. simple molecular graphene segments, into an eclipsed arrangement, separated by 351.64(15) pm, the most precise measure to date of the vdW distance in
AA-stacked graphene bilayers. STM experiments reveal that this cyclophane physisorbs onto nonmagnetic Cu(111) with a face-on geometry, whereas on Co(111) strong chemisorption occurs, with drastic geometric changes to the molecule — a consequence of the drastically altered
electronic coupling between the two pyrene groups? Moreover, spin-polarized STM here detected spin polarization of the ferrocene units of comparable strength, but opposite direction, to that of the pristine Co surface. This showcases the efficiency with which ferrocene transmits spin polarization by the spin-dependent hybridization mechanism, and that covalent linking of ferrocene into the cyclophane architecture allows us to enforce face-on hybridization to ferromagnetic surfaces. For more on this first step in our route to metallocene nanopatterning and engineering spin interfaces, see
Cyclophanes providing two (or more) aromatic π systems are of great interest in molecular spintronics as one aromatic group can strongly chemisorb onto e.g. ferromagnetic metal substrate, leading to magnetic interfacial effects such as magnetic hardening, and imparting spin polarization on the remaining aromatic building blocks. We found that ferrocene pillars are able to lock two pyrene groups, i.e. simple molecular graphene segments, into an eclipsed arrangement, separated by 351.64(15) pm, the most precise measure to date of the vdW distance in
AA-stacked graphene bilayers. STM experiments reveal that this cyclophane physisorbs onto nonmagnetic Cu(111) with a face-on geometry, whereas on Co(111) strong chemisorption occurs, with drastic geometric changes to the molecule — a consequence of the drastically altered
electronic coupling between the two pyrene groups? Moreover, spin-polarized STM here detected spin polarization of the ferrocene units of comparable strength, but opposite direction, to that of the pristine Co surface. This showcases the efficiency with which ferrocene transmits spin polarization by the spin-dependent hybridization mechanism, and that covalent linking of ferrocene into the cyclophane architecture allows us to enforce face-on hybridization to ferromagnetic surfaces. For more on this first step in our route to metallocene nanopatterning and engineering spin interfaces, see
M. Metzelaars, C.M. Schneider, J. Rawson, D.E. Bürgler, S. Sanz, F. Matthes, P. Kögerler, T. Hattori, S. Schleicher, B. Borca, "Cyclophane with Eclipsed Pyrene Units Enables Construction of Spin Interfaces with Chemical Accuracy", Chem. Sci. 2021, 12, 8430-8437. ( doi:10.1039/D1SC01036K). doi:10.1039/D1SC01036K).
Interested in even larger pyrene-ferrocene cyclophanes? See
M. Metzelaars, S. Sanz, J. Rawson, R. Hartmann, C.M. Schneider, P. Kögerler, "Fusing pyrene and ferrocene into a chiral, redox-active triangle", Chem. Commun. 2021, 57, 6660-6663.( doi:10.1039/D1CC02191E). doi:10.1039/D1CC02191E).
SCO Post-functionalization
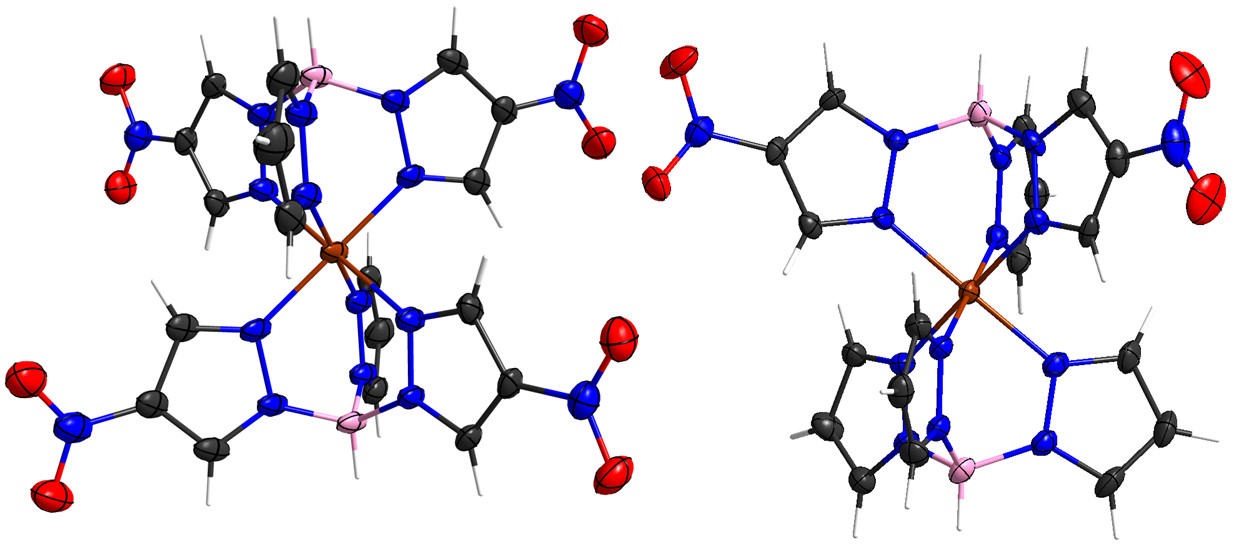 Iron(II) tris(pyrazolyl)borate complexes are a well-established staple in spin crossover chemistry. To enable the covalent integration of such magnetic molecules into various environments, we looked at post-functionalization strategies and saw that simple ligand metathesis gives rise to a surprisingly diverse set of symmetric as well as
asymmetric derivatives. Why has no one ever tried this before? This we do not know, but here you can see what is actually possible with such bona fide building blocks: Iron(II) tris(pyrazolyl)borate complexes are a well-established staple in spin crossover chemistry. To enable the covalent integration of such magnetic molecules into various environments, we looked at post-functionalization strategies and saw that simple ligand metathesis gives rise to a surprisingly diverse set of symmetric as well as
asymmetric derivatives. Why has no one ever tried this before? This we do not know, but here you can see what is actually possible with such bona fide building blocks:
H. Flötotto, T. Secker, P. Kögerler, C. Besson, "Amine-functionalized spin crossover building blocks", Eur. J. Inorg. Chem. 2019, 43, 4621-4624. ( doi:10.1002/ejic.201900916). doi:10.1002/ejic.201900916).
Tellurium Throws Off the Shackles of a Heteroelement
 The traditional role of a heterogroup in heteropolyoxometalates is that of a templating group, for example silicate securing the tetrahedral arrangement of {W3} triads in the classical {PW12} Keggin architecture. However, it is actually possible to emancipate these groups and allow them to assume the functions of addenda atoms, with the same (structural) rights and responsibilities of the early transition metal constituents in classical polyoxometalates. Our synthetic approach resulted in the spectacular 3.5 nm structure of the polyoxotungstotellurite [H4TeIV48WVI78O372]80−, approaching the size of proteins, and demonstrates how to shatter the glass ceiling for hetero:addenda atom ratios. See:
The traditional role of a heterogroup in heteropolyoxometalates is that of a templating group, for example silicate securing the tetrahedral arrangement of {W3} triads in the classical {PW12} Keggin architecture. However, it is actually possible to emancipate these groups and allow them to assume the functions of addenda atoms, with the same (structural) rights and responsibilities of the early transition metal constituents in classical polyoxometalates. Our synthetic approach resulted in the spectacular 3.5 nm structure of the polyoxotungstotellurite [H4TeIV48WVI78O372]80−, approaching the size of proteins, and demonstrates how to shatter the glass ceiling for hetero:addenda atom ratios. See:
J.-X. Liu, N.V. Izarova, P. Kögerler, "Blurring the line between addenda and heteroatoms in a giant polyoxotungstotellurite", Chem. Commun 2019, 55, 10744-10747.
(doi:10.1039/C9CC04623B).
Mechanism of Fulvene Formation
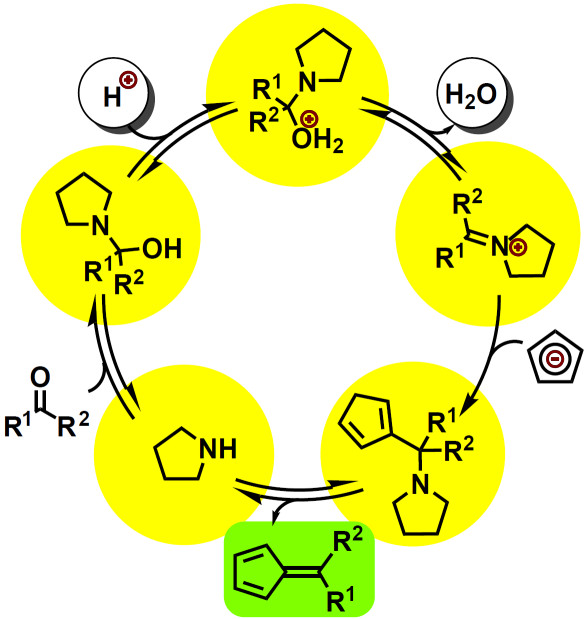 Polycyclopentadienyl ligand systems remain a challenging synthetic target. We recently looked at substituted fulvenes, i.e. suitable precursors for such ligands, and found that their formation can be catalyzed by buffer systems. This greatly expands the scope of products accessible by literature procedures. See our mechanistic study:
Polycyclopentadienyl ligand systems remain a challenging synthetic target. We recently looked at substituted fulvenes, i.e. suitable precursors for such ligands, and found that their formation can be catalyzed by buffer systems. This greatly expands the scope of products accessible by literature procedures. See our mechanistic study:
P. Sieverding, J. Osterbrink, C. Besson, P. Kögerler, "Kinetics and mechanism of pyrrolidine buffer-catalyzed fulvene formation", J. Org. Chem. 2019, 84, 486-494.
(doi:10.1021/acs.joc.8b01660).
PO-LAN-DE: Lanthanide-Borohydride Phases
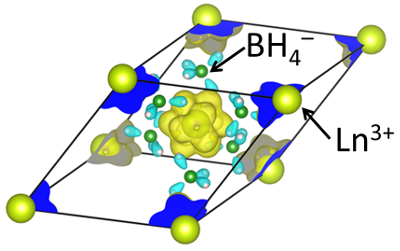 In our collaboration with the Grochala group (Warsaw), we investigated how borohydride, the tamer brother of the hydride anion, can mediate magnetic exchange between lanthanide centers. Interestingly, due to the boron 2p orbitals that are involved in the superexchange, BH4– can transmit both ferromagnetic and antiferromagnetic exchange couplings. Surprisingly, magnetic measurements point to very strong ligand field splitting and, in consequence, significant covalent components in the Ln-H bonds. See
In our collaboration with the Grochala group (Warsaw), we investigated how borohydride, the tamer brother of the hydride anion, can mediate magnetic exchange between lanthanide centers. Interestingly, due to the boron 2p orbitals that are involved in the superexchange, BH4– can transmit both ferromagnetic and antiferromagnetic exchange couplings. Surprisingly, magnetic measurements point to very strong ligand field splitting and, in consequence, significant covalent components in the Ln-H bonds. See
W. Wegner, J. van Leusen, J. Majewski, W. Grochala, P. Kögerler "Borohydride as magnetic superexchange pathway in late lanthanide borohydrides", Eur. J. Inorg. Chem. 2019, 13, 1776-1783 (doi:10.1002/ejic.201801488).
Contacts via Functionalized Polyoxometalate Nanoclusters
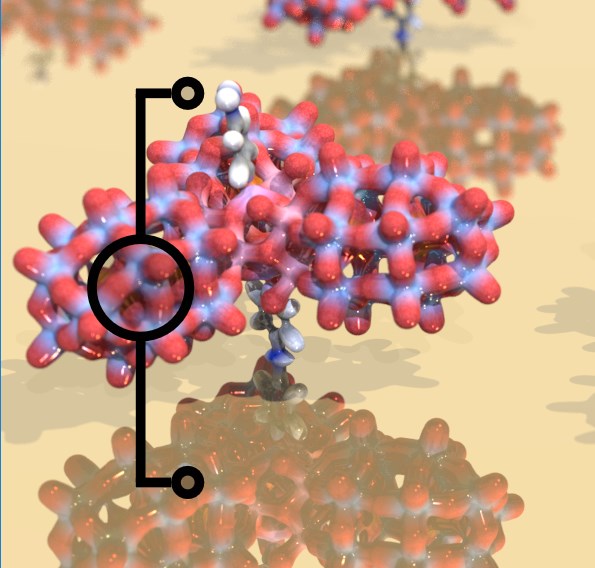 The development of molecular spintronics requires understanding and mastering the contacts between individual molecules and metallic electrodes. A critical factor here is the ability to access the
magnetically relevant molecular orbits of a metal surface-attached molecule, which requires knowledge of their energies with respect to their environments. We approach this tricky problem with specifically functionalized polyoxometalate nanoclusters. This allowed us to pinpoint
the frontier orbital energies, both for the molecule-metal and the molecule-water interface. See
The development of molecular spintronics requires understanding and mastering the contacts between individual molecules and metallic electrodes. A critical factor here is the ability to access the
magnetically relevant molecular orbits of a metal surface-attached molecule, which requires knowledge of their energies with respect to their environments. We approach this tricky problem with specifically functionalized polyoxometalate nanoclusters. This allowed us to pinpoint
the frontier orbital energies, both for the molecule-metal and the molecule-water interface. See
X. Yi, N.V. Izarova, M. Stuckart, D. Guérin, L. Thomas, S. Lenfant, D. Vuillaume, J. van Leusen, T. Duchoň, S. Nemšák, S.D.M. Bourone, S. Schmitz, P. Kögerler "Probing frontier orbital energies of {Co9(P2W15)3} polyoxometalate clusters at molecule–metal and molecule–water interfaces", J. Am. Chem. Soc. 2017, 139, 14501-14510 ( doi:10.1021/jacs.7b07034). doi:10.1021/jacs.7b07034).
Surprise, Surprise: The Electronic Structure of Linear Coordination Compounds with NHC Ligands
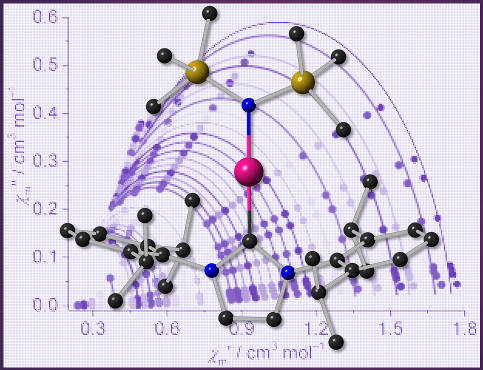 Metal ions in exotic coordination environments represent highly attractive targets in magnetochemistry. We went one step further and looked at under-coordinated (linear) Fe and Co complexes comprising non-innocent ligands. A comparative analysis of the magnetic characteristics, assuming different electron distribution scenarios, revealed Co(II) and Fe(II) centers and NHC radical anions — contrary to the common assumption of M(I) centers in such complexes. See our joint paper with Andreas Danopoulos and Pierre Braunstein in:
Metal ions in exotic coordination environments represent highly attractive targets in magnetochemistry. We went one step further and looked at under-coordinated (linear) Fe and Co complexes comprising non-innocent ligands. A comparative analysis of the magnetic characteristics, assuming different electron distribution scenarios, revealed Co(II) and Fe(II) centers and NHC radical anions — contrary to the common assumption of M(I) centers in such complexes. See our joint paper with Andreas Danopoulos and Pierre Braunstein in:
A. Danopoulos, P. Braunstein, K. Monakhov, J. van Leusen, P. Kögerler, M. Clémancey, J.-M. Latour, A. Benayad, M. Tromp, E. Rezabal, G. Frison, "Heteroleptic, Two-coordinate [M(NHC){N(SiMe3)2}] (M = Co, Fe) Complexes: Synthesis, Reactivity and Magnetism Rationalized by an Unexpected Metal Oxidation State", Dalton Trans. 2017, 46, 1163-1171 (doi:10.1039/C6DT03565E).
True Classical/Non-Classical Polyoxometalate Hybrids: A Radically Novel
Point of Departure Towards Single-Molecule Electronics
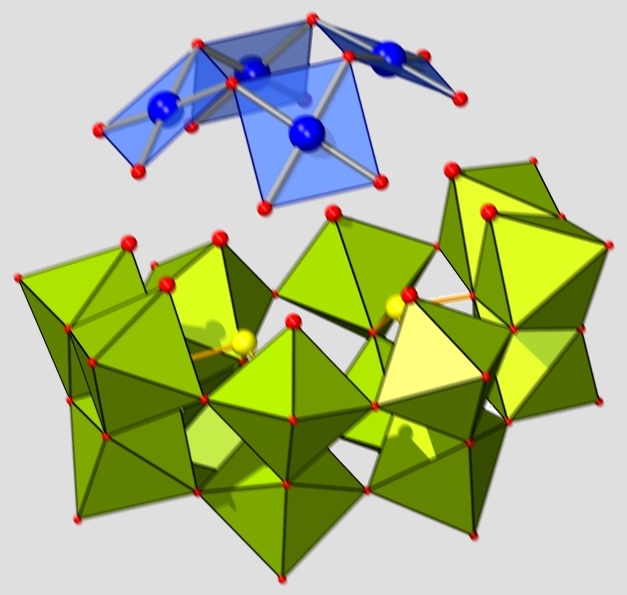 Multiple, precise contacts to a single molecule represents a technological challenge that is far from trivial. As mentioned earlier, we explore the use of hybrid cluster structures to pre-fabricate the crucial molecule-metal interfaces, ideally with atomic precision. In this context we now present model systems in which classical polyoxometalates (comprising early transition metal addenda atoms) and non-conventional POMs (based on late transition metals) are directly joined, without the need for additional support groups — with the prospect of transforming the latter into metallic nano-structures via element-selective reduction. Identifying reaction conditions commensurate for the formation of both classical and non-conventional POMs and their co-condensation has been termed a "holy grail" of molecular metal oxide chemistry, and is described in:
Multiple, precise contacts to a single molecule represents a technological challenge that is far from trivial. As mentioned earlier, we explore the use of hybrid cluster structures to pre-fabricate the crucial molecule-metal interfaces, ideally with atomic precision. In this context we now present model systems in which classical polyoxometalates (comprising early transition metal addenda atoms) and non-conventional POMs (based on late transition metals) are directly joined, without the need for additional support groups — with the prospect of transforming the latter into metallic nano-structures via element-selective reduction. Identifying reaction conditions commensurate for the formation of both classical and non-conventional POMs and their co-condensation has been termed a "holy grail" of molecular metal oxide chemistry, and is described in:
N.V. Izarova, B. Santiago-Schübel, S. Willbold, V. Heß, P. Kögerler, "Classical/Non-classical Polyoxometalate Hybrids",
Chem. Eur. J. 2016, 22, 16052-16056 ( doi:10.1002/chem.201604238). doi:10.1002/chem.201604238).
Congratulations and Welcome to Our Incoming CSC and HEC Fellows!
Six PhD students funded by the CSC and HEC will join the group this fall:
Shengyang Guan, Tuba Iftikhar, Jinxiu Liu, Sidra Sarwar, Waqas Ali Shah, and Wenyan Wang
Magnetism of Berkelium Coordination Compounds
 In comparison to their 4f siblings, the magnetism of actinide ions is highly complex due to the energetic similarity of their single-ion effects. The "full" model Hamiltonian implemented in CONDON enabled, for the first time, the modeling and a precise understanding of the electronic structure of Bk(III) coordination compounds. Such insight will also help improve separation of heavy elements. For this collaboration with Thomas Albrecht-Schmitt (Florida State University) see:
In comparison to their 4f siblings, the magnetism of actinide ions is highly complex due to the energetic similarity of their single-ion effects. The "full" model Hamiltonian implemented in CONDON enabled, for the first time, the modeling and a precise understanding of the electronic structure of Bk(III) coordination compounds. Such insight will also help improve separation of heavy elements. For this collaboration with Thomas Albrecht-Schmitt (Florida State University) see:
M.A. Silver, S.K. Cary, J.A. Johnson, R.E. Baumbach, A.A. Arico, M. Luckey, M. Urban, J.C. Wang, M.J. Polinski, A. Chemey, G. Liu, K.-W. Chen, S.M. Van Cleve, M.L. Marsh, T.M. Eaton, L.J. van de Burgt, A.L. Gray, D.E. Hobart, K. Hanson, L. Maron, F. Gendron, J. Autschbach, M. Speldrich, P. Kögerler, P. Yang, J. Braley, T.E. Albrecht-Schmitt, "Characterization of berkelium(III) dipicolinate and borate compounds in solution and the solid state",
Science 2016, 353, aaf3762 (doi:10.1126/science.aaf3762). Also featured in Chemical & Engineering News.
Magnetism Reveals Bonding Characteristics of 4f and 5f Orbitals
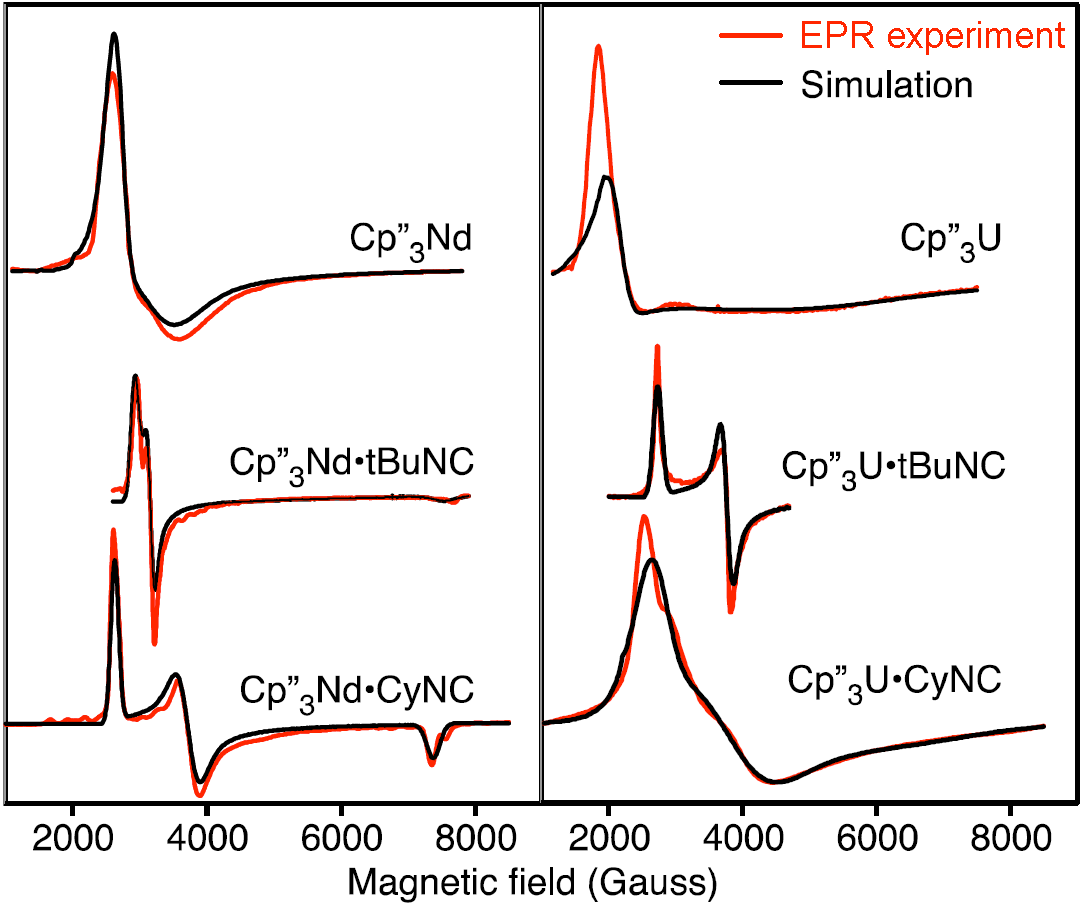 In a comparative study on 4f3 and 5f3 complexes with carefully chosen sets of π- and σ-bonding ligands, spearheaded by Wayne Lukens, we demonstrate that magnetic (susceptibility and EPR spectroscopy) data allows for far-reaching conclusions on the covalent bonding character of the magnetically active f orbitals. See our joint work with several US partner groups:
In a comparative study on 4f3 and 5f3 complexes with carefully chosen sets of π- and σ-bonding ligands, spearheaded by Wayne Lukens, we demonstrate that magnetic (susceptibility and EPR spectroscopy) data allows for far-reaching conclusions on the covalent bonding character of the magnetically active f orbitals. See our joint work with several US partner groups:
W. Lukens, M. Speldrich, P. Yang, T.J. Duignan, J. Autschbach, P. Kögerler, "The roles of 4f and 5f orbitals in bonding: a magnetochemical, crystal field and density functional theory-based study",
Dalton Trans. 2016, 45, 11508-11521 ( doi:10.1039/c6dt00634e). doi:10.1039/c6dt00634e).
A Critical View on Effective Models and ab initio Simulations in Molecular Magnetism
Prompted by the tremendous feedback we have received, we have made this review freely accessible to anyone.
The physical understanding of the microscopic magnetic structure of molecular magnets is often undermined by "effective" approximations that aim to project the quantum mechanical reality of such molecules onto simplified empirical models. While this might lead to deceptively nice-looking fits, the results are often of limited physical meaning, in particular in the case of more anisotropic spin centers. Read more about how such "frankenspin monsters" compare to (more physically sound) "full" model interpretation in:
J. van Leusen, M. Speldrich, H. Schilder, P. Kögerler, "Comprehensive Insight into Molecular Magnetism via CONDON: Full vs. Effective Models",
Coord. Chem. Rev. 2015, 289-290, 137-148 ( doi:10.1016/j.ccr.2014.10.011). doi:10.1016/j.ccr.2014.10.011).
Semimetal Functionalization
... allows us to greatly expand classical polyoxovanadate architectures and to introduce novel physical properties and organic functionalization strategies. We summarize the state-of-the-art of this emerging field of polyoxometalate chemistry in our recent review in:
K.Yu. Monakhov, W. Bensch, P. Kögerler, "Semimetal-functionalised polyoxovanadates", Chem. Soc. Rev. 2015, 44, 8443-8483 ( doi:10.1039/c5cs00531k). doi:10.1039/c5cs00531k).
Open Access Availability
We now aim to provide open access to all of our more recent and upcoming papers. Articles available via "Green" or "Gold" open access models will be marked ( ) in our list of publications. ) in our list of publications.
Congratulations to Kirill!
Dr. Kirill Monakhov has received a prestigious Emmy Noether Fellowship of the Deutsche Forschungsgemeinschaft. His independent Junior Research Group will work on the "Development of Heteropolyvanadate Spin Clusters as Candidates for Future Redox-Based Memory Devices".
Congratulations and Farewell to Claire!
Starting August 2015, Dr. Claire Besson will join the Chemistry Department at George Washington University (Washington, D.C., USA) as a tenure-track assistant professor.
We wish you all the best and a highly successful future!
A Survey on Single-Molecule Magnets based on Polyoxometalates
Polyoxometalates, in particular polyoxomolybdates and -tungstates, provide countless diamagnetic molecular frameworks that we readily can functionalize by integration of spin centers, e.g. paramagnetic transition metal ions. However, only in the past few years have some of these magnetic clusters been identified as single-molecule magnets, meaning that they are characterized by a slowing down of the magnetization relaxation, and possibly even magnetization hysteresis akin to bulk ferromagnets, at low temperatures. We review the current state of affairs in:
N.V. Izarova, P. Kögerler, "Polyoxometalate-based Single-Molecule Magnets", in Trends in Polyoxometalates Research (Eds.: L. Ruhlmann, D. Schaming), Nova Science Publishers, Hauppauge, 2015 ( get PDF). get PDF).
High-Valent Iridium Enters Polyoxometalates: A
Russian-German Collaboration
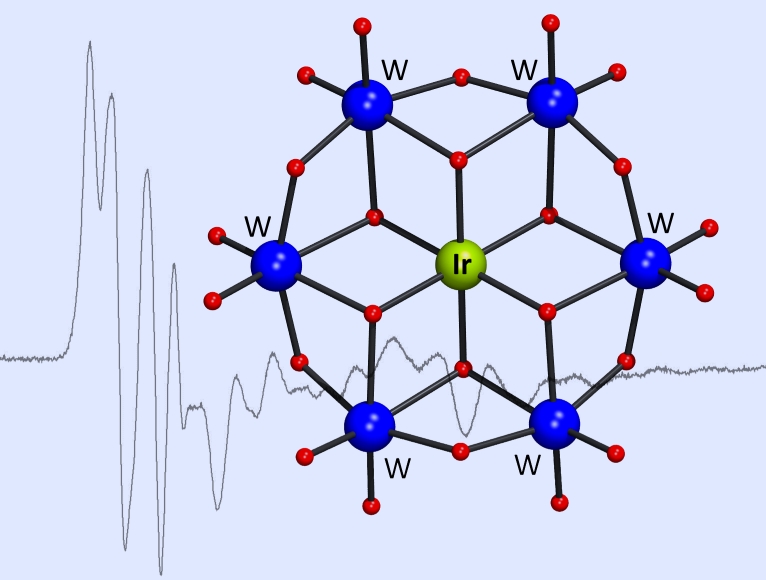 The higher oxidation states of iridium are paramount for the role of Ir complexes as promoters or intermediates in oxidation reactions. Yet, the preparative coordination chemistry of Ir(III) and Ir(IV) is notoriously complicated. Jointly with the group of Maxim Sokolov (Novosibirsk) we reconcile the seemingly incompatible aqueous chemistry of iridium cations and tungstates and provide a convenient blueprint for the development of Ir(IV)-based POMs and possibly even Ir(V)-POM chemistry, which promises intriguing electrochemical and catalytic properties. See:
The higher oxidation states of iridium are paramount for the role of Ir complexes as promoters or intermediates in oxidation reactions. Yet, the preparative coordination chemistry of Ir(III) and Ir(IV) is notoriously complicated. Jointly with the group of Maxim Sokolov (Novosibirsk) we reconcile the seemingly incompatible aqueous chemistry of iridium cations and tungstates and provide a convenient blueprint for the development of Ir(IV)-based POMs and possibly even Ir(V)-POM chemistry, which promises intriguing electrochemical and catalytic properties. See:
S.A. Adonin, N.V. Izarova, C. Besson, P.A. Abramov, B. Santiago-Schübel, P. Kögerler, V.P. Fedin, M.N. Sokolov, "An Ir(IV)-containing
Polyoxometalate",
Chem. Commun. 2015, 51, 1222-1225 ( doi:10.1039/C4CC09271F). doi:10.1039/C4CC09271F).
Hypersensitive Polyoxovanadate Spin Clusters
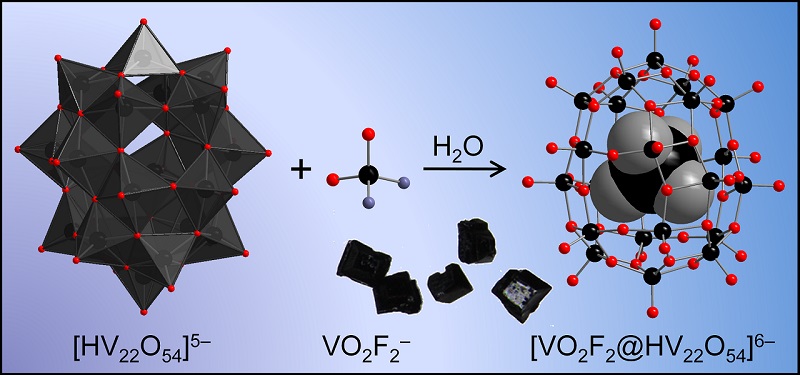 Polyoxovanadate host-guest complexes are known since ages (read: 20 years). Especially mixed-valent species display a pronounced tendency towards structural complementarity, where the shape of the guest determines the host cluster type, despite very weak, purely electrostatic host-guest interactions. A closer look at the {V22O54(X)} family revealed a bunch of surprises linked to the intricate distribution of vanadium 3d electron density and its ultra-sensitive response to perturbations. See: Polyoxovanadate host-guest complexes are known since ages (read: 20 years). Especially mixed-valent species display a pronounced tendency towards structural complementarity, where the shape of the guest determines the host cluster type, despite very weak, purely electrostatic host-guest interactions. A closer look at the {V22O54(X)} family revealed a bunch of surprises linked to the intricate distribution of vanadium 3d electron density and its ultra-sensitive response to perturbations. See:
K.Yu. Monakhov, O. Linnenberg, P. Kozłowski, J. van Leusen, C. Besson, T. Secker, A. Ellern, X. López, J.M. Poblet, P. Kögerler, "Supramolecular Recognition Influences Magnetism in [X@HVIV8VV14O54]6- Self-Assemblies with Symmetry-Breaking Guest Anions",
Chem. Eur. J. 2015, 21, 2387-2397 (doi:10.1002/chem.201403858).
Counting on Neodynium for Molecular Spintronics
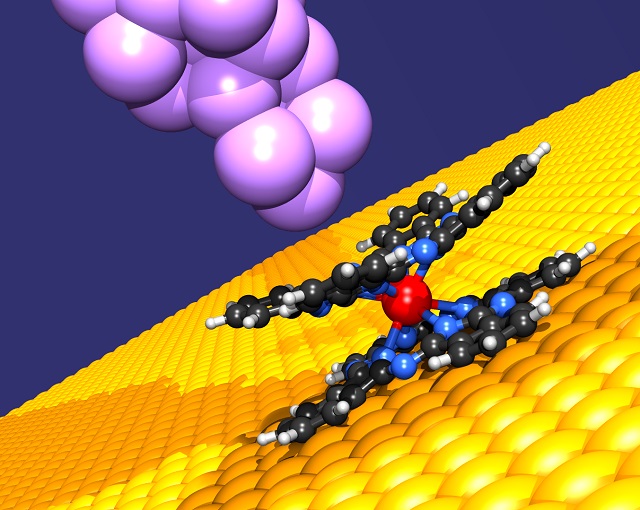 Quantum charge transport through single-molecule magnets is at the heart of molecular spintronics. However, the magnetically active orbitals need to be at the right energy so that they can actually interact with the electrons tunneling through the molecule. We discovered that early lanthanide ions such as Nd(III) fullfil these requirements. This allows, for the first time, the direct observation of molecular 4f-states by scanning tunneling microscopy. See our work in collaboration with our physics partners at Research Center Jülich: Quantum charge transport through single-molecule magnets is at the heart of molecular spintronics. However, the magnetically active orbitals need to be at the right energy so that they can actually interact with the electrons tunneling through the molecule. We discovered that early lanthanide ions such as Nd(III) fullfil these requirements. This allows, for the first time, the direct observation of molecular 4f-states by scanning tunneling microscopy. See our work in collaboration with our physics partners at Research Center Jülich:
S. Fahrendorf, N. Atodiresei, C. Besson, V. Caciuc, F. Matthes, S. Blügel, P. Kögerler, D. E. Bürgler, C. M. Schneider, "Accessing 4f-states in Single-Molecule Spintronics",
Nature Commun. 2013, 4, 2425 ( doi:10.1038/ncomms3425). doi:10.1038/ncomms3425).
This work was also highlighted in Nature Nanotechnology (doi:10.1038/nnano.2013.245).
COST Action "Polyoxometalate Chemistry for Molecular Nanoscience" (PoCheMoN) approved
 Paul Kögerler will act as Vice Chair for the COST Action CM1203 which aims to consolidate and communicate the efforts in polyoxometalate chemistry in 21 European countries as well as several near-neighbor and international partner countries. The EU-funded Action will run until November 2016.
Paul Kögerler will act as Vice Chair for the COST Action CM1203 which aims to consolidate and communicate the efforts in polyoxometalate chemistry in 21 European countries as well as several near-neighbor and international partner countries. The EU-funded Action will run until November 2016.
Stabilization and Control of Fe-Pt Nanoparticles with Polyoxometalates
 Magnetic metal nanoparticles of a few nm fill the gap between the magnetism of single or few metal atoms and cooperative phenomena of condensed phases. Yet, the stabilization and the controlled synthesis of mono-disperese ultra-small nanoparticles (less than 5 nm) remain tricky. Fortunately, functionalized polyoxometalates of the Keggin type can stabilize the surface of such nanoparticles, yielding water-soluble core-shell systems. For our results on such POM-nanoparticle hybrids based on technologically important, hard ferromagnetic FePt phases see:
Magnetic metal nanoparticles of a few nm fill the gap between the magnetism of single or few metal atoms and cooperative phenomena of condensed phases. Yet, the stabilization and the controlled synthesis of mono-disperese ultra-small nanoparticles (less than 5 nm) remain tricky. Fortunately, functionalized polyoxometalates of the Keggin type can stabilize the surface of such nanoparticles, yielding water-soluble core-shell systems. For our results on such POM-nanoparticle hybrids based on technologically important, hard ferromagnetic FePt phases see:
K.M. Seemann, A. Bauer, J. Kindervater, M. Meyer, C. Besson, M. Luysberg, P. Durkin, W. Pyckhout-Hintzen, N. Budisa, R. Georgii, C.M. Schneider, P. Kögerler, "Polyoxometalate-stabilized, water dispersible Fe2Pt magnetic nanoparticles", Nanoscale 2013, 5, 2511-2519 ( doi:10.1039/c3nr33374d). doi:10.1039/c3nr33374d).
Getting Polyoxometalate Nanoreactors to Work
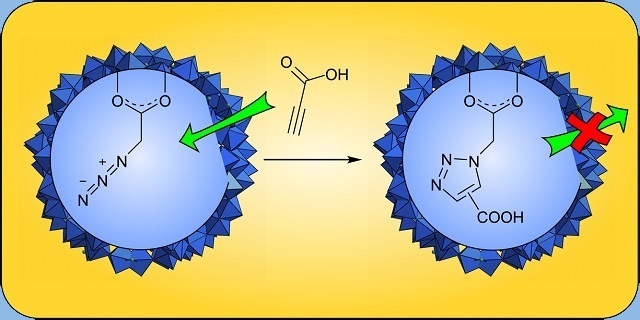 Spherical oxomolybdate clusters of the Keplerate type provide highly defined interior cavities (with a volume of approx. 1.5 nm3) as well as pores. We were able to demonstrate that these systems can indeed function as nanoreactors in which steric confinement translates into specific regioselectivies. At the same time, running an organic cycloaddition reaction within this nanoreactor shields the reactants from catalytically active species in the solution surrounding the Keplerate clusters.
Spherical oxomolybdate clusters of the Keplerate type provide highly defined interior cavities (with a volume of approx. 1.5 nm3) as well as pores. We were able to demonstrate that these systems can indeed function as nanoreactors in which steric confinement translates into specific regioselectivies. At the same time, running an organic cycloaddition reaction within this nanoreactor shields the reactants from catalytically active species in the solution surrounding the Keplerate clusters.
C. Besson, S. Schmitz, K.M. Capella, S. Kopilevich, I.A. Weinstock, P. Kögerler, "A regioselective Huisgen reaction inside a Keplerate polyoxomolybdate nanoreactor", Dalton Trans. 2012, 41, 9852-9854 (doi:10.1039/c2dt30502j).
Prof. Kögerler receives ERC Grant (Consolidator Level) "MOLSPINTRON"
 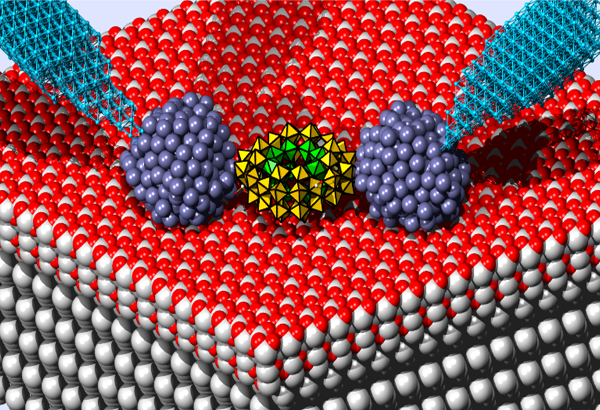 Revolutionary innovations in microelectronics mandate new materials: molecular magnets, used as integral transistor components, are expected to offer several decisive advantages, such as extremely low energy consumption and highly complex switching functions that are hard to realize using conventional semiconductor logic devices. The foundation for these characteristics is the joint utilization of magnetic and electronic quantum states of individual magnetic molecules. Revolutionary innovations in microelectronics mandate new materials: molecular magnets, used as integral transistor components, are expected to offer several decisive advantages, such as extremely low energy consumption and highly complex switching functions that are hard to realize using conventional semiconductor logic devices. The foundation for these characteristics is the joint utilization of magnetic and electronic quantum states of individual magnetic molecules.
In our ERC project 'Synthetic Expansion of Magnetic Molecules Into Spintronic Devices' (11/2012-10/2017), we aim to tackle one of central difficulties in molecular spin electronics: precise and reproducible contacts of the particle. We utilize highly stable magnetic metal oxide nanomolecules and exploit their versatile surface chemistry. Thus, contacts can be made synthetically, i.e., with atomic precision, creating both conductive and nonconductive interfaces.
Alternative Metal Oxide Cluster Chemistry: The Dark Secrets of Cerium
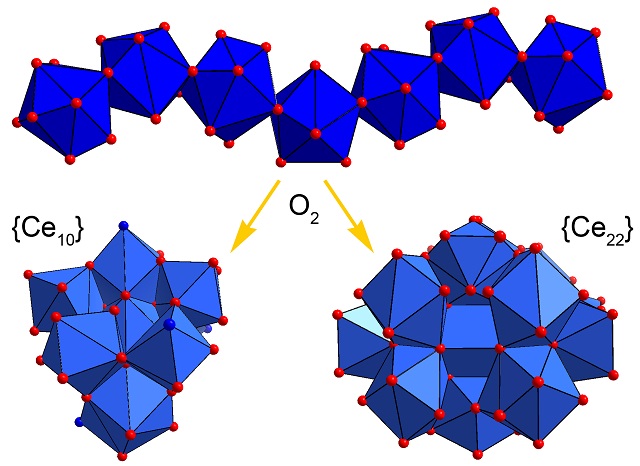 Classical polyoxometalates are typically based on group 5/6 transition metals (V, Nb, Mo, W). What about their communist left-leaning neighbors with less valence electrons? Surprisingly, high-nuclearity molecular cerium oxides can indeed be formed via controlled condensation reactions. Read how their chemistry and structures mimic that of archetypal polyoxometalates in:
Classical polyoxometalates are typically based on group 5/6 transition metals (V, Nb, Mo, W). What about their communist left-leaning neighbors with less valence electrons? Surprisingly, high-nuclearity molecular cerium oxides can indeed be formed via controlled condensation reactions. Read how their chemistry and structures mimic that of archetypal polyoxometalates in:
I.L. Malaestean, A. Ellern, S. Baca, P. Kögerler, "Cerium oxide nanoclusters: Commensurate with concepts of polyoxometalate chemistry?", Chem. Commun. 2012, 48, 1499-1501 (doi:10.1039/c1cc14725k).
|
 Article. Ag(II) as Spin Super-Polarizer in Molecular Spin Clusters. M. Domański, J. van Leusen, M. Metzelaars, P. Kögerler, W. Grochala, J. Phys. Chem. A 2022, 9618-9626. DOI: 10.1021/acs.jpca.2c06032
Article. Ag(II) as Spin Super-Polarizer in Molecular Spin Clusters. M. Domański, J. van Leusen, M. Metzelaars, P. Kögerler, W. Grochala, J. Phys. Chem. A 2022, 9618-9626. DOI: 10.1021/acs.jpca.2c06032
|
 Research Article. Structural Phase Transitions and Magnetic Superexchange in MIAgIIF3 Perovskites at High Pressure. Ł. Wolański, M. Metzelaars, J. van Leusen, P. Kögerler, W. Grochala, Chem. Eur. J. 2022, e202200712. DOI: 10.1002/chem.202200712
Research Article. Structural Phase Transitions and Magnetic Superexchange in MIAgIIF3 Perovskites at High Pressure. Ł. Wolański, M. Metzelaars, J. van Leusen, P. Kögerler, W. Grochala, Chem. Eur. J. 2022, e202200712. DOI: 10.1002/chem.202200712
|
 Full Paper. Magnetic Phase Transitions in a Ni4O4-Cubane-Based Metal–Organic Framework. M.A. Nadeem, M.C.C. Ng, J. van Leusen, P. Kögerler, J.A. Stride, Chem. Eur. J. 2020, 26, 7589-7594. DOI: 10.1002/chem.202000867
Full Paper. Magnetic Phase Transitions in a Ni4O4-Cubane-Based Metal–Organic Framework. M.A. Nadeem, M.C.C. Ng, J. van Leusen, P. Kögerler, J.A. Stride, Chem. Eur. J. 2020, 26, 7589-7594. DOI: 10.1002/chem.202000867
|
 Communication. Amine-Functionalized Spin Crossover Building Blocks. H. Flötotto, T. Secker, P. Kögerler, C. Besson, Eur. J. Inorg. Chem. 2019, 43, 4621-4624. DOI: 10.1002/ejic.201900916
Communication. Amine-Functionalized Spin Crossover Building Blocks. H. Flötotto, T. Secker, P. Kögerler, C. Besson, Eur. J. Inorg. Chem. 2019, 43, 4621-4624. DOI: 10.1002/ejic.201900916
|
 Article. Ion-Directed Coordinative Polymerization of Copper(II) Pyridyl-Alcohol Complexes Through Thiane Functionalities. S. Schmitz, N.V. Izarova, C. Besson, J. van Leusen, P. Kögerler, K.Yu. Monakhov, Z. Anorg. Allg. Chem. 2019, 645, 409-415. DOI: 10.1002/zaac.201800469
Article. Ion-Directed Coordinative Polymerization of Copper(II) Pyridyl-Alcohol Complexes Through Thiane Functionalities. S. Schmitz, N.V. Izarova, C. Besson, J. van Leusen, P. Kögerler, K.Yu. Monakhov, Z. Anorg. Allg. Chem. 2019, 645, 409-415. DOI: 10.1002/zaac.201800469
|
 Article. Probing Frontier Orbital Energies of {Co9(P2W15)3} Polyoxometalate Clusters at Molecule-Metal and Molecule-Water Interfaces. X. Yi, N.V. Izarova, M. Stuckart, D. Guérin, L. Thomas, S. Lenfant, D. Vuillaume, J. van Leusen, T. Duchoň, S. Nemšák, S.D.M. Bourone, S. Schmitz, P. Kögerler, J. Am. Chem. Soc. 2017, 139, 14501-14510. DOI: 10.1021/jacs.7b07034
Article. Probing Frontier Orbital Energies of {Co9(P2W15)3} Polyoxometalate Clusters at Molecule-Metal and Molecule-Water Interfaces. X. Yi, N.V. Izarova, M. Stuckart, D. Guérin, L. Thomas, S. Lenfant, D. Vuillaume, J. van Leusen, T. Duchoň, S. Nemšák, S.D.M. Bourone, S. Schmitz, P. Kögerler, J. Am. Chem. Soc. 2017, 139, 14501-14510. DOI: 10.1021/jacs.7b07034
|
 Full Paper. Heterometallic [Cu2Ln3] (Ln = DyIII, GdIII and HoIII) and [Cu4Ln2] (Ln = DyIII and HoIII) Compounds: Synthesis, Structure, and Magnetism. S. Biswas, P. Bag, S. Das, S. Kundu, J. van Leusen, P. Kögerler, V. Chandrasekhar, Eur. J. Inorg. Chem. 2017, 8, 1129-1142. DOI: 10.1002/ejic.201601210
Full Paper. Heterometallic [Cu2Ln3] (Ln = DyIII, GdIII and HoIII) and [Cu4Ln2] (Ln = DyIII and HoIII) Compounds: Synthesis, Structure, and Magnetism. S. Biswas, P. Bag, S. Das, S. Kundu, J. van Leusen, P. Kögerler, V. Chandrasekhar, Eur. J. Inorg. Chem. 2017, 8, 1129-1142. DOI: 10.1002/ejic.201601210
|
 Review Article. Semimetal-functionalised polyoxovanadates. K.Yu. Monakhov, W. Bensch, P. Kögerler, Chem. Soc. Rev. 2015, 44, 8443-8483. DOI: 10.1039/C5CS00531K
Review Article. Semimetal-functionalised polyoxovanadates. K.Yu. Monakhov, W. Bensch, P. Kögerler, Chem. Soc. Rev. 2015, 44, 8443-8483. DOI: 10.1039/C5CS00531K
|
 Full Paper. Supramolecular Recognition Influences Magnetism in [X@HVIV8VV14O54]6- Self-Assemblies with Symmetry-Breaking Guest Anions. K.Yu. Monakhov, O. Linnenberg, P. Kozłowski, J. van Leusen, C. Besson, T. Secker, A. Ellern, X. López, J.M. Poblet, P. Kögerler, Chem. Eur. J. 2015, 21, 2387-2397. DOI: 10.1002/chem.201403858
Full Paper. Supramolecular Recognition Influences Magnetism in [X@HVIV8VV14O54]6- Self-Assemblies with Symmetry-Breaking Guest Anions. K.Yu. Monakhov, O. Linnenberg, P. Kozłowski, J. van Leusen, C. Besson, T. Secker, A. Ellern, X. López, J.M. Poblet, P. Kögerler, Chem. Eur. J. 2015, 21, 2387-2397. DOI: 10.1002/chem.201403858
|
 Communication. A polyoxometalate-based single-molecule magnet with an S = 21/2 ground state. X. Fang, P. Kögerler, M. Speldrich, H. Schilder, M. Luban, Chem. Commun. 2012, 48, 1218-1220. DOI: 10.1039/C1CC15520B
Communication. A polyoxometalate-based single-molecule magnet with an S = 21/2 ground state. X. Fang, P. Kögerler, M. Speldrich, H. Schilder, M. Luban, Chem. Commun. 2012, 48, 1218-1220. DOI: 10.1039/C1CC15520B
|
 Perspective. Structure-related frustrated magnetism of nanosized polyoxometalates: aesthetics and properties in harmony. P. Kögerler, B. Tsukerblat, A. Müller, Dalton Trans. 2010, 39, 21-36. DOI: 10.1039/B910716A
Perspective. Structure-related frustrated magnetism of nanosized polyoxometalates: aesthetics and properties in harmony. P. Kögerler, B. Tsukerblat, A. Müller, Dalton Trans. 2010, 39, 21-36. DOI: 10.1039/B910716A
|
 Communication. Acetate-driven polyoxometalate demetalation: An open-shell diiron polytungstate comprising two rotational Keggin isomers. B. Botar, P. Kögerler, Dalton Trans. 2008, 3150-3152. DOI: 10.1039/B802147C
Communication. Acetate-driven polyoxometalate demetalation: An open-shell diiron polytungstate comprising two rotational Keggin isomers. B. Botar, P. Kögerler, Dalton Trans. 2008, 3150-3152. DOI: 10.1039/B802147C
|
 Communication. Structural and Compositional Control in {M12} Cobalt and Nickel Coordination Clusters Detected Magnetochemically and with Cryospray Mass Spectrometry. G. J. T. Cooper, G. N. Newton, P. Kögerler, D.-L. Long, L. Engelhardt, M. Luban, L. Cronin, Angew. Chem. Int. Ed. 2007, 46, 1340-1344. DOI: 10.1002/anie.200603870
Communication. Structural and Compositional Control in {M12} Cobalt and Nickel Coordination Clusters Detected Magnetochemically and with Cryospray Mass Spectrometry. G. J. T. Cooper, G. N. Newton, P. Kögerler, D.-L. Long, L. Engelhardt, M. Luban, L. Cronin, Angew. Chem. Int. Ed. 2007, 46, 1340-1344. DOI: 10.1002/anie.200603870
|
|
 In October 2022, Paul Kögerler was elected to serve as Dean of Student Affairs of RWTH's Faculty of Mathematics, Computer Science and Natural Sciences.
In October 2022, Paul Kögerler was elected to serve as Dean of Student Affairs of RWTH's Faculty of Mathematics, Computer Science and Natural Sciences.
 With their high magnetic moments (up to ca. 11 μB for Dy3+ or Ho3+) and their strong spin-orbit coupling, certain lanthanide ions should be highly attractive components of molecular magnetic materials and also molecular spintronics. At the same time, their spin-bearing 4f orbitals are contracted (read: teeny-weeny), which limits their overlap with the frontier orbitals of bridging ligands and, thus, the intramolecular magnetic exchange interactions. In turn, their highly interesting magnetic phenomena manifest only at very low temperatures. In the binational project POL-LAN-DE, funded by NCN (PL) and DFG (DE), our friends from Warsaw and us explored how the magnetic polarisation introduced by electron-hungry Ag2+ cations can salvage this situation. The Ag2+ cation has a 4d9 configuration and a 4d electron hole with an extremely high electron affinity of close to 11 eV, leading a pronounced hybridization with the valence orbitals of surrounding ligands. This induces a strong spin polarization even on rock-hard fluoride bridging ligands and can greatly increase the magnetic coupling in Ln-F-Ln bridges. For a systematic study, see
With their high magnetic moments (up to ca. 11 μB for Dy3+ or Ho3+) and their strong spin-orbit coupling, certain lanthanide ions should be highly attractive components of molecular magnetic materials and also molecular spintronics. At the same time, their spin-bearing 4f orbitals are contracted (read: teeny-weeny), which limits their overlap with the frontier orbitals of bridging ligands and, thus, the intramolecular magnetic exchange interactions. In turn, their highly interesting magnetic phenomena manifest only at very low temperatures. In the binational project POL-LAN-DE, funded by NCN (PL) and DFG (DE), our friends from Warsaw and us explored how the magnetic polarisation introduced by electron-hungry Ag2+ cations can salvage this situation. The Ag2+ cation has a 4d9 configuration and a 4d electron hole with an extremely high electron affinity of close to 11 eV, leading a pronounced hybridization with the valence orbitals of surrounding ligands. This induces a strong spin polarization even on rock-hard fluoride bridging ligands and can greatly increase the magnetic coupling in Ln-F-Ln bridges. For a systematic study, see Cyclophanes providing two (or more) aromatic π systems are of great interest in molecular spintronics as one aromatic group can strongly chemisorb onto e.g. ferromagnetic metal substrate, leading to magnetic interfacial effects such as magnetic hardening, and imparting spin polarization on the remaining aromatic building blocks. We found that ferrocene pillars are able to lock two pyrene groups, i.e. simple molecular graphene segments, into an eclipsed arrangement, separated by 351.64(15) pm, the most precise measure to date of the vdW distance in
AA-stacked graphene bilayers. STM experiments reveal that this cyclophane physisorbs onto nonmagnetic Cu(111) with a face-on geometry, whereas on Co(111) strong chemisorption occurs, with drastic geometric changes to the molecule — a consequence of the drastically altered
electronic coupling between the two pyrene groups? Moreover, spin-polarized STM here detected spin polarization of the ferrocene units of comparable strength, but opposite direction, to that of the pristine Co surface. This showcases the efficiency with which ferrocene transmits spin polarization by the spin-dependent hybridization mechanism, and that covalent linking of ferrocene into the cyclophane architecture allows us to enforce face-on hybridization to ferromagnetic surfaces. For more on this first step in our route to metallocene nanopatterning and engineering spin interfaces, see
Cyclophanes providing two (or more) aromatic π systems are of great interest in molecular spintronics as one aromatic group can strongly chemisorb onto e.g. ferromagnetic metal substrate, leading to magnetic interfacial effects such as magnetic hardening, and imparting spin polarization on the remaining aromatic building blocks. We found that ferrocene pillars are able to lock two pyrene groups, i.e. simple molecular graphene segments, into an eclipsed arrangement, separated by 351.64(15) pm, the most precise measure to date of the vdW distance in
AA-stacked graphene bilayers. STM experiments reveal that this cyclophane physisorbs onto nonmagnetic Cu(111) with a face-on geometry, whereas on Co(111) strong chemisorption occurs, with drastic geometric changes to the molecule — a consequence of the drastically altered
electronic coupling between the two pyrene groups? Moreover, spin-polarized STM here detected spin polarization of the ferrocene units of comparable strength, but opposite direction, to that of the pristine Co surface. This showcases the efficiency with which ferrocene transmits spin polarization by the spin-dependent hybridization mechanism, and that covalent linking of ferrocene into the cyclophane architecture allows us to enforce face-on hybridization to ferromagnetic surfaces. For more on this first step in our route to metallocene nanopatterning and engineering spin interfaces, see Iron(II) tris(pyrazolyl)borate complexes are a well-established staple in spin crossover chemistry. To enable the covalent integration of such magnetic molecules into various environments, we looked at post-functionalization strategies and saw that simple ligand metathesis gives rise to a surprisingly diverse set of symmetric as well as
asymmetric derivatives. Why has no one ever tried this before? This we do not know, but here you can see what is actually possible with such bona fide building blocks:
Iron(II) tris(pyrazolyl)borate complexes are a well-established staple in spin crossover chemistry. To enable the covalent integration of such magnetic molecules into various environments, we looked at post-functionalization strategies and saw that simple ligand metathesis gives rise to a surprisingly diverse set of symmetric as well as
asymmetric derivatives. Why has no one ever tried this before? This we do not know, but here you can see what is actually possible with such bona fide building blocks: The traditional role of a heterogroup in heteropolyoxometalates is that of a templating group, for example silicate securing the tetrahedral arrangement of {W3} triads in the classical {PW12} Keggin architecture. However, it is actually possible to emancipate these groups and allow them to assume the functions of addenda atoms, with the same (structural) rights and responsibilities of the early transition metal constituents in classical polyoxometalates. Our synthetic approach resulted in the spectacular 3.5 nm structure of the polyoxotungstotellurite [H4TeIV48WVI78O372]80−, approaching the size of proteins, and demonstrates how to shatter the glass ceiling for hetero:addenda atom ratios. See:
The traditional role of a heterogroup in heteropolyoxometalates is that of a templating group, for example silicate securing the tetrahedral arrangement of {W3} triads in the classical {PW12} Keggin architecture. However, it is actually possible to emancipate these groups and allow them to assume the functions of addenda atoms, with the same (structural) rights and responsibilities of the early transition metal constituents in classical polyoxometalates. Our synthetic approach resulted in the spectacular 3.5 nm structure of the polyoxotungstotellurite [H4TeIV48WVI78O372]80−, approaching the size of proteins, and demonstrates how to shatter the glass ceiling for hetero:addenda atom ratios. See: Polycyclopentadienyl ligand systems remain a challenging synthetic target. We recently looked at substituted fulvenes, i.e. suitable precursors for such ligands, and found that their formation can be catalyzed by buffer systems. This greatly expands the scope of products accessible by literature procedures. See our mechanistic study:
Polycyclopentadienyl ligand systems remain a challenging synthetic target. We recently looked at substituted fulvenes, i.e. suitable precursors for such ligands, and found that their formation can be catalyzed by buffer systems. This greatly expands the scope of products accessible by literature procedures. See our mechanistic study: In our collaboration with the Grochala group (Warsaw), we investigated how borohydride, the tamer brother of the hydride anion, can mediate magnetic exchange between lanthanide centers. Interestingly, due to the boron 2p orbitals that are involved in the superexchange, BH4– can transmit both ferromagnetic and antiferromagnetic exchange couplings. Surprisingly, magnetic measurements point to very strong ligand field splitting and, in consequence, significant covalent components in the Ln-H bonds. See
In our collaboration with the Grochala group (Warsaw), we investigated how borohydride, the tamer brother of the hydride anion, can mediate magnetic exchange between lanthanide centers. Interestingly, due to the boron 2p orbitals that are involved in the superexchange, BH4– can transmit both ferromagnetic and antiferromagnetic exchange couplings. Surprisingly, magnetic measurements point to very strong ligand field splitting and, in consequence, significant covalent components in the Ln-H bonds. See The development of molecular spintronics requires understanding and mastering the contacts between individual molecules and metallic electrodes. A critical factor here is the ability to access the
magnetically relevant molecular orbits of a metal surface-attached molecule, which requires knowledge of their energies with respect to their environments. We approach this tricky problem with specifically functionalized polyoxometalate nanoclusters. This allowed us to pinpoint
the frontier orbital energies, both for the molecule-metal and the molecule-water interface. See
The development of molecular spintronics requires understanding and mastering the contacts between individual molecules and metallic electrodes. A critical factor here is the ability to access the
magnetically relevant molecular orbits of a metal surface-attached molecule, which requires knowledge of their energies with respect to their environments. We approach this tricky problem with specifically functionalized polyoxometalate nanoclusters. This allowed us to pinpoint
the frontier orbital energies, both for the molecule-metal and the molecule-water interface. See Metal ions in exotic coordination environments represent highly attractive targets in magnetochemistry. We went one step further and looked at under-coordinated (linear) Fe and Co complexes comprising non-innocent ligands. A comparative analysis of the magnetic characteristics, assuming different electron distribution scenarios, revealed Co(II) and Fe(II) centers and NHC radical anions — contrary to the common assumption of M(I) centers in such complexes. See our joint paper with Andreas Danopoulos and Pierre Braunstein in:
Metal ions in exotic coordination environments represent highly attractive targets in magnetochemistry. We went one step further and looked at under-coordinated (linear) Fe and Co complexes comprising non-innocent ligands. A comparative analysis of the magnetic characteristics, assuming different electron distribution scenarios, revealed Co(II) and Fe(II) centers and NHC radical anions — contrary to the common assumption of M(I) centers in such complexes. See our joint paper with Andreas Danopoulos and Pierre Braunstein in: Multiple, precise contacts to a single molecule represents a technological challenge that is far from trivial. As mentioned
Multiple, precise contacts to a single molecule represents a technological challenge that is far from trivial. As mentioned  In comparison to their 4f siblings, the magnetism of actinide ions is highly complex due to the energetic similarity of their single-ion effects. The "full" model Hamiltonian implemented in CONDON enabled, for the first time, the modeling and a precise understanding of the electronic structure of Bk(III) coordination compounds. Such insight will also help improve separation of heavy elements. For this collaboration with Thomas Albrecht-Schmitt (Florida State University) see:
In comparison to their 4f siblings, the magnetism of actinide ions is highly complex due to the energetic similarity of their single-ion effects. The "full" model Hamiltonian implemented in CONDON enabled, for the first time, the modeling and a precise understanding of the electronic structure of Bk(III) coordination compounds. Such insight will also help improve separation of heavy elements. For this collaboration with Thomas Albrecht-Schmitt (Florida State University) see: In a comparative study on 4f3 and 5f3 complexes with carefully chosen sets of π- and σ-bonding ligands, spearheaded by Wayne Lukens, we demonstrate that magnetic (susceptibility and EPR spectroscopy) data allows for far-reaching conclusions on the covalent bonding character of the magnetically active f orbitals. See our joint work with several US partner groups:
In a comparative study on 4f3 and 5f3 complexes with carefully chosen sets of π- and σ-bonding ligands, spearheaded by Wayne Lukens, we demonstrate that magnetic (susceptibility and EPR spectroscopy) data allows for far-reaching conclusions on the covalent bonding character of the magnetically active f orbitals. See our joint work with several US partner groups:
 The higher oxidation states of iridium are paramount for the role of Ir complexes as promoters or intermediates in oxidation reactions. Yet, the preparative coordination chemistry of Ir(III) and Ir(IV) is notoriously complicated. Jointly with the group of Maxim Sokolov (Novosibirsk) we reconcile the seemingly incompatible aqueous chemistry of iridium cations and tungstates and provide a convenient blueprint for the development of Ir(IV)-based POMs and possibly even Ir(V)-POM chemistry, which promises intriguing electrochemical and catalytic properties. See:
The higher oxidation states of iridium are paramount for the role of Ir complexes as promoters or intermediates in oxidation reactions. Yet, the preparative coordination chemistry of Ir(III) and Ir(IV) is notoriously complicated. Jointly with the group of Maxim Sokolov (Novosibirsk) we reconcile the seemingly incompatible aqueous chemistry of iridium cations and tungstates and provide a convenient blueprint for the development of Ir(IV)-based POMs and possibly even Ir(V)-POM chemistry, which promises intriguing electrochemical and catalytic properties. See: Polyoxovanadate host-guest complexes are known since ages (read: 20 years). Especially mixed-valent species display a pronounced tendency towards structural complementarity, where the shape of the guest determines the host cluster type, despite very weak, purely electrostatic host-guest interactions. A closer look at the {V22O54(X)} family revealed a bunch of surprises linked to the intricate distribution of vanadium 3d electron density and its ultra-sensitive response to perturbations. See:
Polyoxovanadate host-guest complexes are known since ages (read: 20 years). Especially mixed-valent species display a pronounced tendency towards structural complementarity, where the shape of the guest determines the host cluster type, despite very weak, purely electrostatic host-guest interactions. A closer look at the {V22O54(X)} family revealed a bunch of surprises linked to the intricate distribution of vanadium 3d electron density and its ultra-sensitive response to perturbations. See: Quantum charge transport through single-molecule magnets is at the heart of molecular spintronics. However, the magnetically active orbitals need to be at the right energy so that they can actually interact with the electrons tunneling through the molecule. We discovered that early lanthanide ions such as Nd(III) fullfil these requirements. This allows, for the first time, the direct observation of molecular 4f-states by scanning tunneling microscopy. See our work in collaboration with our physics partners at Research Center Jülich:
Quantum charge transport through single-molecule magnets is at the heart of molecular spintronics. However, the magnetically active orbitals need to be at the right energy so that they can actually interact with the electrons tunneling through the molecule. We discovered that early lanthanide ions such as Nd(III) fullfil these requirements. This allows, for the first time, the direct observation of molecular 4f-states by scanning tunneling microscopy. See our work in collaboration with our physics partners at Research Center Jülich: Paul Kögerler will act as Vice Chair for the
Paul Kögerler will act as Vice Chair for the  Magnetic metal nanoparticles of a few nm fill the gap between the magnetism of single or few metal atoms and cooperative phenomena of condensed phases. Yet, the stabilization and the controlled synthesis of mono-disperese ultra-small nanoparticles (less than 5 nm) remain tricky. Fortunately, functionalized polyoxometalates of the Keggin type can stabilize the surface of such nanoparticles, yielding water-soluble core-shell systems. For our results on such POM-nanoparticle hybrids based on technologically important, hard ferromagnetic FePt phases see:
Magnetic metal nanoparticles of a few nm fill the gap between the magnetism of single or few metal atoms and cooperative phenomena of condensed phases. Yet, the stabilization and the controlled synthesis of mono-disperese ultra-small nanoparticles (less than 5 nm) remain tricky. Fortunately, functionalized polyoxometalates of the Keggin type can stabilize the surface of such nanoparticles, yielding water-soluble core-shell systems. For our results on such POM-nanoparticle hybrids based on technologically important, hard ferromagnetic FePt phases see: Spherical oxomolybdate clusters of the Keplerate type provide highly defined interior cavities (with a volume of approx. 1.5 nm3) as well as pores. We were able to demonstrate that these systems can indeed function as nanoreactors in which steric confinement translates into specific regioselectivies. At the same time, running an organic cycloaddition reaction within this nanoreactor shields the reactants from catalytically active species in the solution surrounding the Keplerate clusters.
Spherical oxomolybdate clusters of the Keplerate type provide highly defined interior cavities (with a volume of approx. 1.5 nm3) as well as pores. We were able to demonstrate that these systems can indeed function as nanoreactors in which steric confinement translates into specific regioselectivies. At the same time, running an organic cycloaddition reaction within this nanoreactor shields the reactants from catalytically active species in the solution surrounding the Keplerate clusters.
 Revolutionary innovations in microelectronics mandate new materials: molecular magnets, used as integral transistor components, are expected to offer several decisive advantages, such as extremely low energy consumption and highly complex switching functions that are hard to realize using conventional semiconductor logic devices. The foundation for these characteristics is the joint utilization of magnetic and electronic quantum states of individual magnetic molecules.
Revolutionary innovations in microelectronics mandate new materials: molecular magnets, used as integral transistor components, are expected to offer several decisive advantages, such as extremely low energy consumption and highly complex switching functions that are hard to realize using conventional semiconductor logic devices. The foundation for these characteristics is the joint utilization of magnetic and electronic quantum states of individual magnetic molecules. Classical polyoxometalates are typically based on group 5/6 transition metals (V, Nb, Mo, W). What about their communist left-leaning neighbors with less valence electrons? Surprisingly, high-nuclearity molecular cerium oxides can indeed be formed via controlled condensation reactions. Read how their chemistry and structures mimic that of archetypal polyoxometalates in:
Classical polyoxometalates are typically based on group 5/6 transition metals (V, Nb, Mo, W). What about their communist left-leaning neighbors with less valence electrons? Surprisingly, high-nuclearity molecular cerium oxides can indeed be formed via controlled condensation reactions. Read how their chemistry and structures mimic that of archetypal polyoxometalates in: Article. Ag(II) as Spin Super-Polarizer in Molecular Spin Clusters. M. Domański, J. van Leusen, M. Metzelaars, P. Kögerler, W. Grochala, J. Phys. Chem. A 2022, 9618-9626. DOI: 10.1021/acs.jpca.2c06032
Article. Ag(II) as Spin Super-Polarizer in Molecular Spin Clusters. M. Domański, J. van Leusen, M. Metzelaars, P. Kögerler, W. Grochala, J. Phys. Chem. A 2022, 9618-9626. DOI: 10.1021/acs.jpca.2c06032
 Research Article. Structural Phase Transitions and Magnetic Superexchange in MIAgIIF3 Perovskites at High Pressure. Ł. Wolański, M. Metzelaars, J. van Leusen, P. Kögerler, W. Grochala, Chem. Eur. J. 2022, e202200712. DOI: 10.1002/chem.202200712
Research Article. Structural Phase Transitions and Magnetic Superexchange in MIAgIIF3 Perovskites at High Pressure. Ł. Wolański, M. Metzelaars, J. van Leusen, P. Kögerler, W. Grochala, Chem. Eur. J. 2022, e202200712. DOI: 10.1002/chem.202200712
 Full Paper. Magnetic Phase Transitions in a Ni4O4-Cubane-Based Metal–Organic Framework. M.A. Nadeem, M.C.C. Ng, J. van Leusen, P. Kögerler, J.A. Stride, Chem. Eur. J. 2020, 26, 7589-7594. DOI: 10.1002/chem.202000867
Full Paper. Magnetic Phase Transitions in a Ni4O4-Cubane-Based Metal–Organic Framework. M.A. Nadeem, M.C.C. Ng, J. van Leusen, P. Kögerler, J.A. Stride, Chem. Eur. J. 2020, 26, 7589-7594. DOI: 10.1002/chem.202000867
 Communication. Amine-Functionalized Spin Crossover Building Blocks. H. Flötotto, T. Secker, P. Kögerler, C. Besson, Eur. J. Inorg. Chem. 2019, 43, 4621-4624. DOI: 10.1002/ejic.201900916
Communication. Amine-Functionalized Spin Crossover Building Blocks. H. Flötotto, T. Secker, P. Kögerler, C. Besson, Eur. J. Inorg. Chem. 2019, 43, 4621-4624. DOI: 10.1002/ejic.201900916
 Article. Ion-Directed Coordinative Polymerization of Copper(II) Pyridyl-Alcohol Complexes Through Thiane Functionalities. S. Schmitz, N.V. Izarova, C. Besson, J. van Leusen, P. Kögerler, K.Yu. Monakhov, Z. Anorg. Allg. Chem. 2019, 645, 409-415. DOI: 10.1002/zaac.201800469
Article. Ion-Directed Coordinative Polymerization of Copper(II) Pyridyl-Alcohol Complexes Through Thiane Functionalities. S. Schmitz, N.V. Izarova, C. Besson, J. van Leusen, P. Kögerler, K.Yu. Monakhov, Z. Anorg. Allg. Chem. 2019, 645, 409-415. DOI: 10.1002/zaac.201800469
 Article. Probing Frontier Orbital Energies of {Co9(P2W15)3} Polyoxometalate Clusters at Molecule-Metal and Molecule-Water Interfaces. X. Yi, N.V. Izarova, M. Stuckart, D. Guérin, L. Thomas, S. Lenfant, D. Vuillaume, J. van Leusen, T. Duchoň, S. Nemšák, S.D.M. Bourone, S. Schmitz, P. Kögerler, J. Am. Chem. Soc. 2017, 139, 14501-14510. DOI: 10.1021/jacs.7b07034
Article. Probing Frontier Orbital Energies of {Co9(P2W15)3} Polyoxometalate Clusters at Molecule-Metal and Molecule-Water Interfaces. X. Yi, N.V. Izarova, M. Stuckart, D. Guérin, L. Thomas, S. Lenfant, D. Vuillaume, J. van Leusen, T. Duchoň, S. Nemšák, S.D.M. Bourone, S. Schmitz, P. Kögerler, J. Am. Chem. Soc. 2017, 139, 14501-14510. DOI: 10.1021/jacs.7b07034
 Full Paper. Heterometallic [Cu2Ln3] (Ln = DyIII, GdIII and HoIII) and [Cu4Ln2] (Ln = DyIII and HoIII) Compounds: Synthesis, Structure, and Magnetism. S. Biswas, P. Bag, S. Das, S. Kundu, J. van Leusen, P. Kögerler, V. Chandrasekhar, Eur. J. Inorg. Chem. 2017, 8, 1129-1142. DOI: 10.1002/ejic.201601210
Full Paper. Heterometallic [Cu2Ln3] (Ln = DyIII, GdIII and HoIII) and [Cu4Ln2] (Ln = DyIII and HoIII) Compounds: Synthesis, Structure, and Magnetism. S. Biswas, P. Bag, S. Das, S. Kundu, J. van Leusen, P. Kögerler, V. Chandrasekhar, Eur. J. Inorg. Chem. 2017, 8, 1129-1142. DOI: 10.1002/ejic.201601210
 Review Article. Semimetal-functionalised polyoxovanadates. K.Yu. Monakhov, W. Bensch, P. Kögerler, Chem. Soc. Rev. 2015, 44, 8443-8483. DOI: 10.1039/C5CS00531K
Review Article. Semimetal-functionalised polyoxovanadates. K.Yu. Monakhov, W. Bensch, P. Kögerler, Chem. Soc. Rev. 2015, 44, 8443-8483. DOI: 10.1039/C5CS00531K
 Full Paper. Supramolecular Recognition Influences Magnetism in [X@HVIV8VV14O54]6- Self-Assemblies with Symmetry-Breaking Guest Anions. K.Yu. Monakhov, O. Linnenberg, P. Kozłowski, J. van Leusen, C. Besson, T. Secker, A. Ellern, X. López, J.M. Poblet, P. Kögerler, Chem. Eur. J. 2015, 21, 2387-2397. DOI: 10.1002/chem.201403858
Full Paper. Supramolecular Recognition Influences Magnetism in [X@HVIV8VV14O54]6- Self-Assemblies with Symmetry-Breaking Guest Anions. K.Yu. Monakhov, O. Linnenberg, P. Kozłowski, J. van Leusen, C. Besson, T. Secker, A. Ellern, X. López, J.M. Poblet, P. Kögerler, Chem. Eur. J. 2015, 21, 2387-2397. DOI: 10.1002/chem.201403858
 Communication. A polyoxometalate-based single-molecule magnet with an S = 21/2 ground state. X. Fang, P. Kögerler, M. Speldrich, H. Schilder, M. Luban, Chem. Commun. 2012, 48, 1218-1220. DOI: 10.1039/C1CC15520B
Communication. A polyoxometalate-based single-molecule magnet with an S = 21/2 ground state. X. Fang, P. Kögerler, M. Speldrich, H. Schilder, M. Luban, Chem. Commun. 2012, 48, 1218-1220. DOI: 10.1039/C1CC15520B
 Perspective. Structure-related frustrated magnetism of nanosized polyoxometalates: aesthetics and properties in harmony. P. Kögerler, B. Tsukerblat, A. Müller, Dalton Trans. 2010, 39, 21-36. DOI: 10.1039/B910716A
Perspective. Structure-related frustrated magnetism of nanosized polyoxometalates: aesthetics and properties in harmony. P. Kögerler, B. Tsukerblat, A. Müller, Dalton Trans. 2010, 39, 21-36. DOI: 10.1039/B910716A
 Communication. Acetate-driven polyoxometalate demetalation: An open-shell diiron polytungstate comprising two rotational Keggin isomers. B. Botar, P. Kögerler, Dalton Trans. 2008, 3150-3152. DOI: 10.1039/B802147C
Communication. Acetate-driven polyoxometalate demetalation: An open-shell diiron polytungstate comprising two rotational Keggin isomers. B. Botar, P. Kögerler, Dalton Trans. 2008, 3150-3152. DOI: 10.1039/B802147C
 Communication. Structural and Compositional Control in {M12} Cobalt and Nickel Coordination Clusters Detected Magnetochemically and with Cryospray Mass Spectrometry. G. J. T. Cooper, G. N. Newton, P. Kögerler, D.-L. Long, L. Engelhardt, M. Luban, L. Cronin, Angew. Chem. Int. Ed. 2007, 46, 1340-1344. DOI: 10.1002/anie.200603870
Communication. Structural and Compositional Control in {M12} Cobalt and Nickel Coordination Clusters Detected Magnetochemically and with Cryospray Mass Spectrometry. G. J. T. Cooper, G. N. Newton, P. Kögerler, D.-L. Long, L. Engelhardt, M. Luban, L. Cronin, Angew. Chem. Int. Ed. 2007, 46, 1340-1344. DOI: 10.1002/anie.200603870